Fluorite Processing Plant: The Ultimate Guide to Acid Grade Fluorspar Flotation
Fluorite (also known commercially as Fluorspar) is the primary source of fluorine for the global chemical industry. From the hydrofluoric acid used to manufacture solar panels and lithium-ion battery electrolytes, to the flux required for steel and aluminum smelting, fluorite is an indispensable strategic mineral.
However, turning raw fluorite ore into a highly profitable commercial product is an intricate metallurgical challenge. Fluorite (CaF2) is almost always found entangled with gangue minerals that share terrifyingly similar physical and chemical properties—specifically Calcite (CaCO3) and Barite (BaSO4). Because they all share alkaline earth metal cations (like Calcium), traditional physical separation is often insufficient, and conventional flotation collectors cannot easily distinguish between them.
As a leading global EPC (Engineering, Procurement, and Construction) contractor, OreSolution specializes in decoding these complex mineralogical puzzles. This ultimate engineering guide will dissect the Fluorite Ore Production Line, detailing the "Grind-Float-Regrind" strategies, the critical reagent chemistry needed to separate fluorite from calcite, and how to consistently achieve the coveted 97% "Acid Grade" purity.
The profitability of a fluorite processing plant is heavily skewed towards high purity. While "Metallurgical Grade" (80-85% CaF2) is relatively easy to achieve and sells for a modest price, "Acid Grade" (97%+ CaF2) commands a massive premium. The entire design of a modern fluorite plant must be engineered toward reaching that 97% threshold.
Part 1: Understanding Commercial Fluorspar Grades
Before designing the comminution and flotation circuits, you must define the end product. The fluorite market is rigidly segmented into three primary grades based on Calcium Fluoride (CaF2) content and strict limits on impurities like silica (SiO2), calcium carbonate (CaCO3), and sulfur.
Part 2: The Two Pillars of Fluorite Beneficiation
Depending on the nature of the deposit and the target grade, a fluorite processing plant will utilize one or both of these core technologies:
1. Gravity Separation (For Coarse, Metallurgical Grade)
If the fluorite is coarsely disseminated (large crystals) and your goal is to produce Metspar, gravity separation is highly efficient and inexpensive. Fluorite has a specific gravity of 3.18, while quartz is 2.65. After crushing to an appropriate size (e.g., -10mm), the ore is fed into a Sawtooth Wave Jig. The pulsating water separates the heavier fluorite chunks from the lighter silica rock.
2. Froth Flotation (For Fine, Acid Grade)
To produce Acid Grade fluorspar, or to process finely disseminated ores where the fluorite is intimately locked with gangue, gravity separation fails. You must grind the ore to a fine powder and use Froth Flotation. This process relies on manipulating the surface chemistry of the minerals so that air bubbles selectively float the fluorite while leaving the calcite and silica behind.
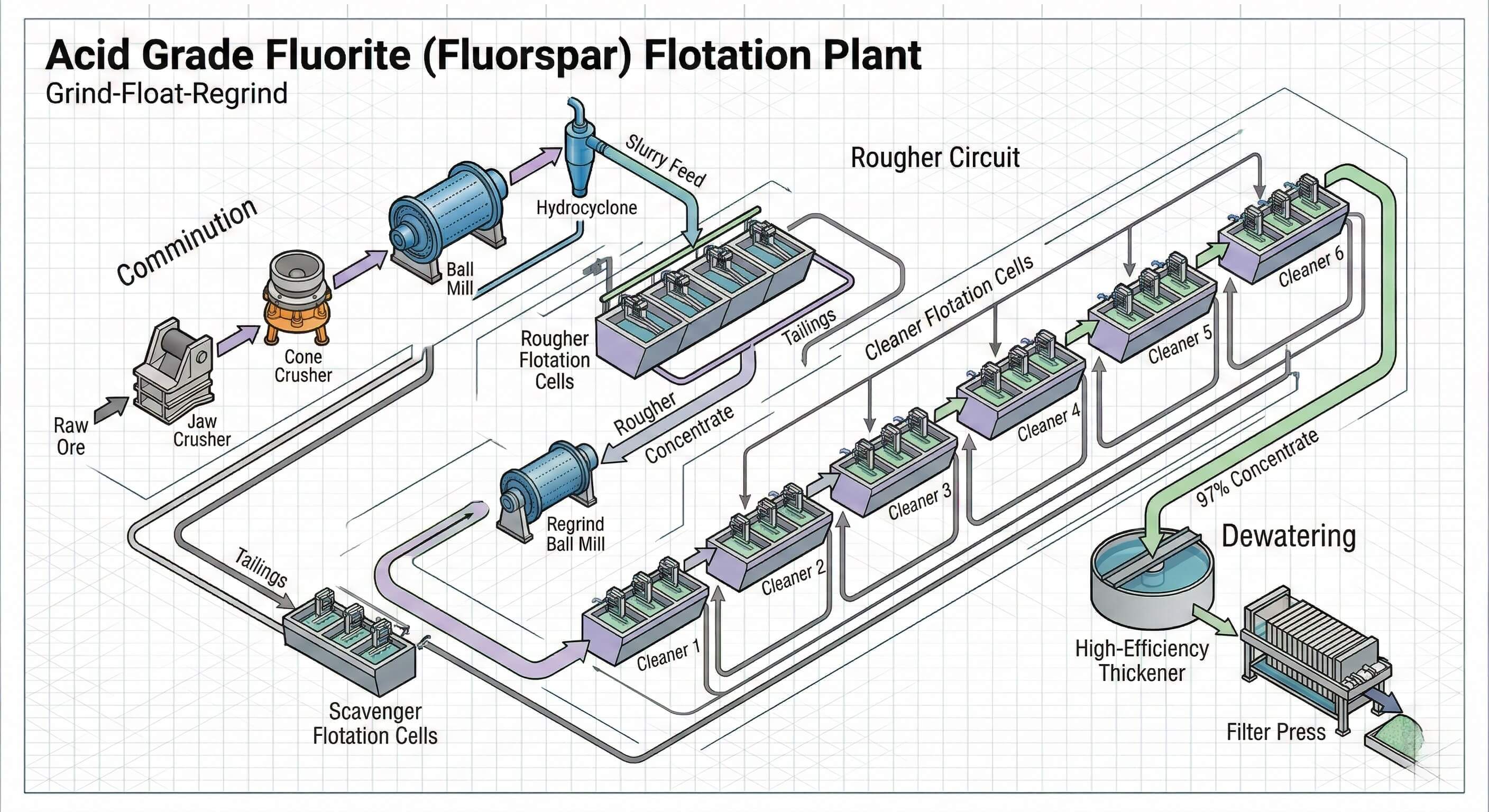
Part 3: Comminution - The "Grind-Float-Regrind" Strategy
The cardinal sin in fluorite flotation is over-grinding. Fluorite is relatively soft and brittle. If you grind the entire ore body down to a fine powder in one step to liberate every last particle, you will create excessive "slimes" (ultra-fine mud). Slimes consume massive amounts of expensive reagents and coat the larger fluorite particles, completely ruining the flotation process.
OreSolution's Approach: We employ a stage-grinding strategy.
- Primary Grinding: After passing through Jaw and Cone Crushers, the ore enters a Ball Mill operating in closed circuit with a Hydrocyclone. The goal is a relatively coarse primary grind—just fine enough to liberate the easiest fluorite particles.
- Rougher Flotation: We immediately float this coarsely ground material to recover a large portion of the fluorite early.
- Regrinding the Concentrate: The rougher concentrate (which still contains locked particles of fluorite and silica/calcite) is sent to a secondary, smaller regrind mill. This highly targeted grinding ensures complete liberation without over-grinding the entire mass of the ore.
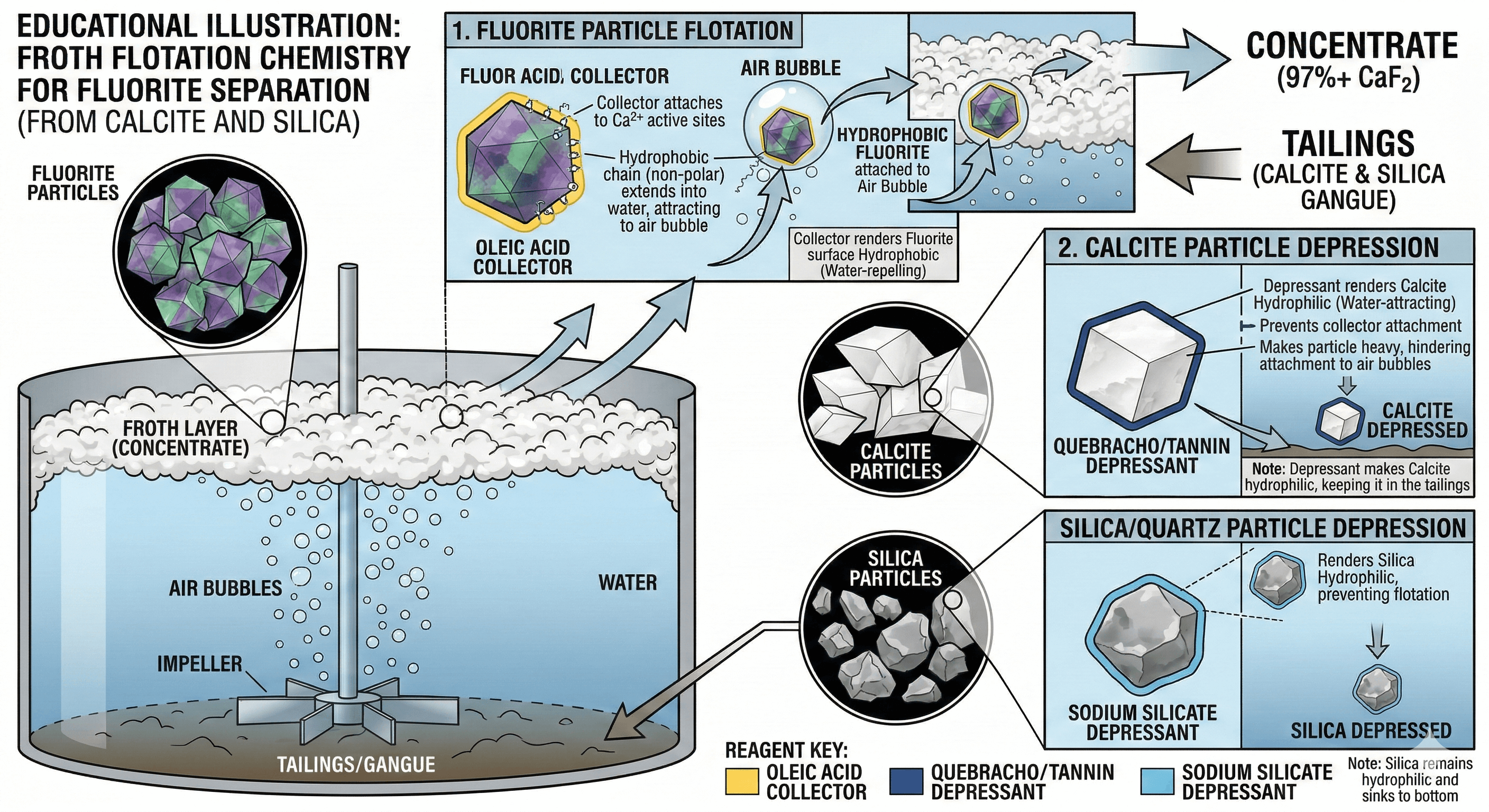
Part 4: The Chemistry of Fluorite Flotation
The success of an Acid Grade fluorite processing plant hinges entirely on the reagent regime. The difficulty lies in the fact that Fluorite (CaF2), Calcite (CaCO3), and Apatite all share the Calcium cation. A standard fatty acid collector (like Oleic Acid) will eagerly attach to all of them, floating the valuable fluorite right alongside the useless calcite.
To achieve separation, we must use highly specific depressants to "blind" the calcite and silica, preventing them from floating.
Part 5: The "Many Cleaners" Flowsheet Architecture
Because the separation between fluorite and its gangue is so delicate, you cannot achieve 97% purity in one or two steps. A standard Acid Grade fluorspar plant features a very long flotation circuit.
- Roughing: To capture as much fluorite as possible.
- Scavenging: Treating the rougher tailings to catch any escaped fluorite before it goes to the dam.
- Cleaning (The Marathon): To push the grade from ~60% up to >97%, the concentrate must pass through a series of Air-Inflated Flotation Cells. It is common for an OreSolution-designed plant to feature 6 to 8 stages of cleaning. In each stage, more depressants are carefully metered in to progressively squeeze out the remaining silica and calcite.
Part 6: Dealing with Complex Polymetallic Fluorite Ores
Often, fluorite is found associated with valuable sulfide minerals, such as Galena (Lead) or Sphalerite (Zinc). These sulfides must be removed before the fluorite flotation circuit, as they will contaminate the final acid-grade product.
The Sequential Strategy:
- First, use Xanthate collectors to float the Lead and Zinc (which can be sold as separate, valuable concentrates).
- The tailings from the Lead/Zinc circuit now become the feed for the Fluorite circuit. We then switch to fatty acid collectors to float the fluorite.
Part 7: Dewatering and Product Handling
The final Acid Grade concentrate emerges from the flotation cells as a wet, frothy slurry. This must be transformed into a dry, shippable powder.
The slurry is first pumped to a High-Efficiency Thickener, which concentrates the solids. The thickened underflow is then pumped at high pressure into a Plate and Frame Filter Press, producing a firm filter cake. For certain markets, this cake must be further processed through a rotary dryer to reduce moisture to below 1% before being bagged.
FAQ: Expert Troubleshooting for Fluorite Plants
A: High silica usually indicates one of two problems: 1) Insufficient liberation: The fluorite is still physically attached to quartz particles. You need to increase the grind time in your regrind mill. 2) Insufficient depression: Your Sodium Silicate (Water Glass) dosage is too low, allowing free quartz to float. Try adjusting the dosage or using an acidified sodium silicate mix for stronger depression.
A: This is the classic fluorite flotation problem. Both minerals respond to the Oleic Acid collector. You must refine your depressant strategy. Ensure your pH is strictly controlled with Sodium Carbonate, and consider adding specialized depressants like Quebracho extract or a precisely mixed ratio of Sodium Silicate and Alum to selectively blind the calcite.
A: Oleic acid is a powerful collector but also acts as a strong frother. In cold water, fatty acids like Oleic acid do not disperse well and can create a tough, sticky froth. Heating the slurry (to around 30°C - 40°C) significantly improves the dispersion of the collector, leading to a more manageable froth and much higher selectivity.
A: Generally, no. Gravity separation (Jigs or Tables) is excellent for producing Metallurgical Grade (+60% CaF2) from coarse ores. However, because gangue minerals are often locked inside the fluorite crystals, producing 97% Acid Grade requires fine grinding (to liberate the impurities), and at fine sizes, gravity separation becomes ineffective. Flotation is required.
Conclusion: Engineering Purity
Designing a fluorite processing plant capable of consistently producing Acid Grade fluorspar is a high-wire act of chemistry and comminution. A poorly designed flowsheet that over-grinds the ore or fails to depress calcite will leave you stuck with low-value Metallurgical Grade product, severely impacting your ROI.
At OreSolution, we eliminate the guesswork. From conducting rigorous bench-scale flotation tests to determine the exact reagent recipe for your specific ore, to designing multi-stage Fluorite Production Lines and supplying the heavy machinery, our EPC turnkey service ensures your plant achieves maximum purity and maximum profit.
Are you developing a fluorite deposit? Contact OreSolution today to consult with our senior metallurgists and begin designing your Acid Grade processing plant.