Scheelite Processing Plant: The Ultimate Guide to Tungsten Flotation & Beneficiation
Tungsten (W) is a strategic metal renowned for possessing the highest melting point of all metallic elements. It is indispensable in aerospace, military applications, and the manufacturing of ultra-hard cemented carbides. While historically derived from Wolframite (black tungsten), the rapid depletion of those reserves has forced the global mining industry to pivot aggressively towards Scheelite (CaWO4)—the "white tungsten."
However, extracting Scheelite is a metallurgical nightmare. Unlike Wolframite, which is heavy and magnetic, Scheelite is typically finely disseminated and non-magnetic, making gravity separation insufficient for achieving commercial grades (typically >65% WO3). The industry standard is Froth Flotation, but Scheelite flotation is notoriously complex due to its intimate association with other calcium-bearing gangue minerals.
As a leading global EPC (Engineering, Procurement, and Construction) contractor, OreSolution specializes in designing high-recovery Scheelite Production Lines. This comprehensive engineering guide will decode the extreme chemistry required to separate Scheelite from Calcite and Fluorite, exploring both the traditional "Petrov Heating Process" and modern room-temperature techniques.
The central paradox of scheelite beneficiation is the "Calcium Conundrum." Scheelite (CaWO4), Calcite (CaCO3), Fluorite (CaF2), and Apatite [Ca5(PO4)3(F,Cl,OH)] all share the same active surface cation: Calcium (Ca2+). A standard fatty acid collector will indiscriminately attach to all of them, floating the valuable tungsten right alongside the useless waste. Precision chemistry is the only way to break this bond.
Part 1: The Mineralogical Battlefield
Before designing a scheelite processing plant, a detailed mineralogical analysis is mandatory. The flowsheet is entirely dictated by which specific calcium minerals are present in the ore body.
Part 2: Comminution - Avoiding Scheelite Slimes
Scheelite is incredibly brittle (friable). Like its cousin Wolframite, if it is over-ground, it turns into microscopic "slimes" (-10 microns). Once Scheelite becomes slime, it loses its floatability and is washed away into the tailings dam.
The OreSolution Comminution Strategy:
- Stage Grinding: We never attempt to reach liberation size in a single pass. The ore is crushed using Jaw Crushers and Cone Crushers before entering the mill.
- Rod Mills over Ball Mills: For primary grinding, a Rod Mill is vastly superior. Its "line-contact" grinding action shatters the harder quartz while minimizing the over-grinding of the softer Scheelite.
- Strict Classification: The mill operates in a closed circuit. Oversize material is returned, while liberated particles are immediately rushed to the flotation circuit to prevent further attrition.
Part 3: Pre-Treatment - The Essential Sulfide Float
Most Scheelite deposits (especially skarn deposits) are heavily contaminated with sulfide minerals like Pyrite, Pyrrhotite, Molybdenite, or Bismuthinite. Sulfides are a fatal poison to the downstream Tungsten smelting process.
Before any Scheelite flotation begins, the slurry must undergo a Bulk Sulfide Flotation step. Using inexpensive Xanthate collectors, all the sulfide minerals are floated off into a separate concentrate (which can often be sold for its copper, molybdenum, or bismuth value). The "tailings" from this sulfide circuit—now free of sulfur—become the clean feed for the main Scheelite circuit.
Part 4: Scheelite Rougher Flotation (The Bulk Catch)
With the sulfides removed, the goal of the Rougher circuit is maximum recovery. We want to float the Scheelite, but because of the "Calcium Conundrum," we accept that Calcite, Fluorite, and Apatite will float alongside it.
The result of this stage is a low-grade Bulk Rougher Concentrate (typically 5% - 15% WO3), representing over 85% of the total tungsten recovery.
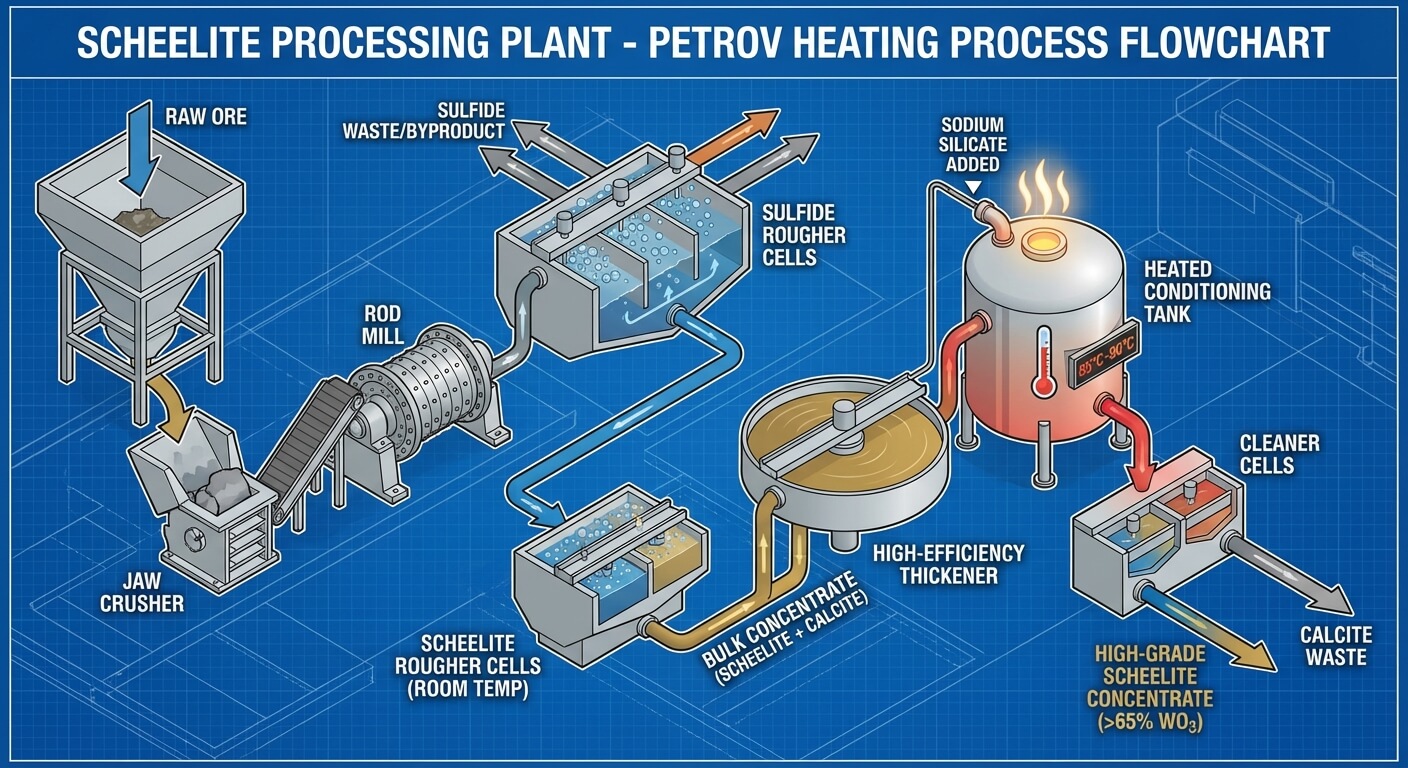
Part 5: Scheelite Cleaner Flotation - The Heating Process (Petrov)
The Bulk Rougher Concentrate is full of Calcite and Fluorite. Standard flotation at room temperature struggles to separate them. The globally recognized solution, and a hallmark of a high-end scheelite processing plant, is the Petrov Process (Heating Flotation).
This process exploits a unique chemical vulnerability in the gangue minerals when exposed to extreme heat and high concentrations of Sodium Silicate.
- Thickening: The dilute Bulk Concentrate is pumped to a High-Efficiency Thickener to increase the solid density to about 60%.
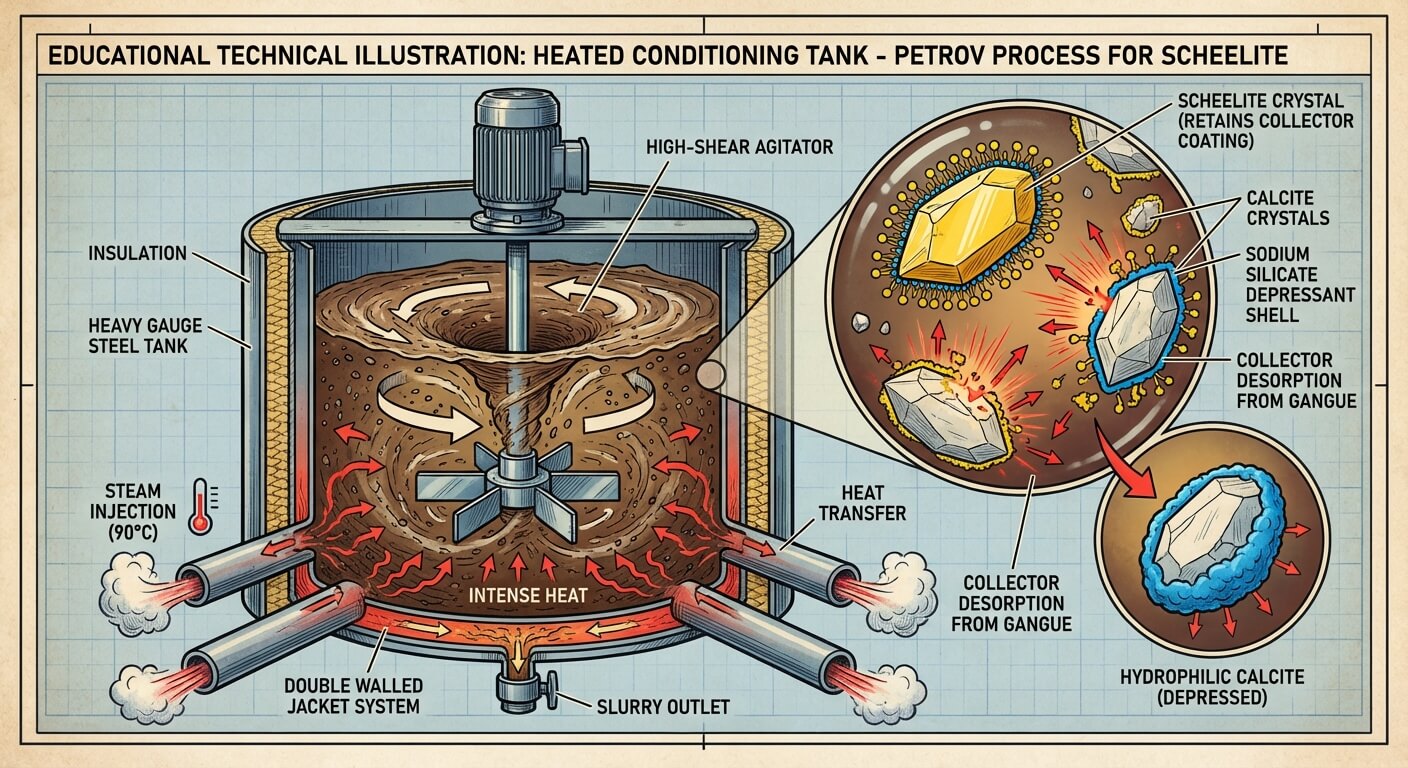
- High-Shear Heating (The Crucial Step): The thick slurry is pumped into specialized, heavily insulated Heating Conditioning Tanks. Steam is injected to heat the slurry to 85°C - 90°C (185°F - 195°F). A massive dose of Sodium Silicate (Water Glass) is added, and the mixture is intensely agitated for 1 to 2 hours.
- The Desorption Mechanism: The intense heat and mechanical shear physically tear the collector (Oleic Acid) off the surfaces of the Calcite and Fluorite. The high concentration of Sodium Silicate then rapidly coats these now-bare gangue minerals, permanently depressing them. In stark contrast, the collector bond on the Scheelite is thermodynamically stable and survives the boiling process.
- Dilution and Cleaning: The boiling slurry is diluted with cold water and fed into a series of Cleaner Flotation Cells. Now, only the Scheelite floats. The Calcite and Fluorite sink and are rejected as tailings.
By repeating this cleaner step 3 to 5 times, the grade is pushed from 10% WO3 up to a premium commercial grade of >65% WO3.
Part 6: Dewatering the Final Concentrate
The final, high-grade Scheelite froth must be dewatered before shipping to the APT (Ammonium Paratungstate) refinery.
The concentrate is first thickened in a smaller Thickener, then pumped at high pressure into a Plate and Frame Filter Press or a vacuum disc filter. The resulting filter cake is dried in a Rotary Dryer, reducing moisture to below 1%, resulting in a fine, heavy, whitish powder ready for market.
FAQ: Troubleshooting Scheelite Flotation Plants
A: High Phosphorus indicates that Apatite (a calcium phosphate mineral) is surviving the Heating Process and floating with the Scheelite. To fix this, you must refine your depressant regime. During the Heating (Petrov) stage, carefully test the ratio of Sodium Silicate to your collector. In severe cases, an acid leaching step (using Hydrochloric acid) on the final concentrate may be required to dissolve the Apatite.
A: Yes, "Room Temperature" or "Normal Temperature" Scheelite flotation is possible and saves massive amounts of energy (steam) costs. However, it requires highly sophisticated, often proprietary, customized collector/depressant combinations (e.g., specific Chelating collectors mixed with acidified Sodium Silicate). OreSolution's metallurgical lab can determine if your specific ore body is amenable to room-temperature separation.
A: The most likely culprit is "Slimes" or "Hard Water." If you over-ground the ore, the ultra-fine slimes will coat the Scheelite, blinding it from the collector. If your process water contains high levels of free Calcium or Magnesium ions (hard water), these ions will react with the Oleic acid to form insoluble "calcium soaps," essentially destroying the collector before it can attach to the ore. You must soften the water using Sodium Carbonate (Soda Ash) before adding the collector.
Conclusion: The OreSolution EPC Advantage
Designing a profitable scheelite processing plant is one of the ultimate tests of metallurgical engineering. A flowsheet that ignores the nuance of the "Calcium Conundrum" will inevitably produce a useless mixed concentrate of tungsten, calcite, and fluorite, leading to complete project failure.
At OreSolution, we eliminate this risk. From conducting exhaustive bench-scale heating flotation tests in our laboratory to engineering the robust steam-jacketed conditioning tanks and high-capacity flotation cells required for the Petrov Process, we deliver turnkey Scheelite Production Lines that guarantee the 65% WO3 grade your buyers demand.
Are you developing a complex Scheelite skarn deposit? Contact OreSolution today to consult with our senior process engineers and design an optimized, high-recovery beneficiation plant.




