Copper Flotation Process: The Ultimate Guide to Copper-Lead-Zinc Separation
Driven by the global transition to electric vehicles (EVs), renewable energy grids, and advanced electronics, Copper is rapidly becoming the most critical metal of the 21st century. However, the mining industry faces a severe challenge: easy-to-process, high-grade copper deposits are largely exhausted. Today, miners are forced to develop deep, low-grade polymetallic ores—complex rocks where Copper (Chalcopyrite), Lead (Galena), and Zinc (Sphalerite) are tightly intergrown with massive amounts of iron pyrite and silica.
Separating these metals into distinct, high-purity commercial concentrates is considered one of the most difficult engineering feats in mineral processing. If you fail to separate the zinc from the copper concentrate, the smelter will hit you with massive financial penalties. If you over-grind the lead, it turns into unrecoverable slime, washing millions of dollars into the tailings dam.
As a leading global EPC (Engineering, Procurement, and Construction) contractor, OreSolution specializes in designing highly efficient Copper Ore Beneficiation Production Lines and Lead-Zinc Processing Plants. This definitive guide decodes the intricate chemistry, machinery, and flowsheet design required for successful polymetallic froth flotation.
In polymetallic processing, grade and recovery are always inversely proportional. Pushing for 95% copper recovery often results in a low-grade concentrate contaminated with lead and zinc. The art of copper flotation plant design lies in finding the exact economic "sweet spot" tailored to your specific smelter contract.
Part 1: The Challenge of Polymetallic Ore Mineralogy

Before selecting a single Flotation Machine, you must understand the microscopic battlefield of your ore. In a typical Copper-Lead-Zinc (Cu-Pb-Zn) deposit, the minerals are locked together like pieces of a microscopic puzzle.
- Chalcopyrite (CuFeS2): The primary copper mineral. It floats easily but is often intimately associated with iron pyrite.
- Galena (PbS): The primary lead mineral. Extremely heavy and brittle. It tends to over-grind easily, turning into unrecoverable slimes if the grinding circuit is poorly designed.
- Sphalerite (ZnS): The primary zinc mineral. Naturally, it does not float well with standard collectors. It requires "activation" using Copper Sulfate (CuSO4) before it can be recovered.
- Pyrite (FeS2): The enemy. Iron sulfide has no economic value in this context but consumes expensive reagents and dilutes the concentrate if allowed to float.
The entire copper flotation process relies on manipulating the surface chemistry of these minerals so that air bubbles selectively attach to one mineral (e.g., Copper) while ignoring the others (Lead, Zinc, and Pyrite).
Part 2: Comminution - The Crucial Grinding Strategy
Flotation is a surface phenomenon. If a copper particle is still encased inside a quartz rock, the chemical reagents cannot touch it, and the air bubble cannot lift it. The ore must be crushed and ground until the valuable minerals are physically separated from the waste rock (a state known as liberation).
The Risk of Over-Grinding Galena
The standard comminution circuit starts with a Jaw Crusher, followed by a Cone Crusher, reducing the rock to -12mm. This is fed into a Ball Mill operating in a closed circuit with hydrocyclones.
Here lies the danger: Galena (Lead) is very soft, while Quartz (gangue) is hard. If you grind the ore long enough to liberate the tough quartz, you will pulverize the Galena into ultra-fine mud (slimes). These slimes coat the copper and zinc particles, ruining the entire flotation process.
OreSolution's Engineering Fix: We implement a "Grind-Float-Regrind" strategy. We perform a coarse primary grind to liberate the easily accessible copper and lead, float them out immediately, and then send the remaining coarse tailings to a secondary regrind mill to liberate the more stubborn interlocked particles.
Part 3: Flowsheet Design - Sequential vs Bulk Flotation
Once the ore is liberated as a liquid slurry, how do we separate three different valuable metals? Metallurgists must choose between two primary flowsheet architectures.
In modern lead zinc separation plants handling complex ores, Bulk Flotation followed by Cu-Pb Separation is the most common and economically viable route.
Part 4: Flotation Reagents - The Chemical Symphony
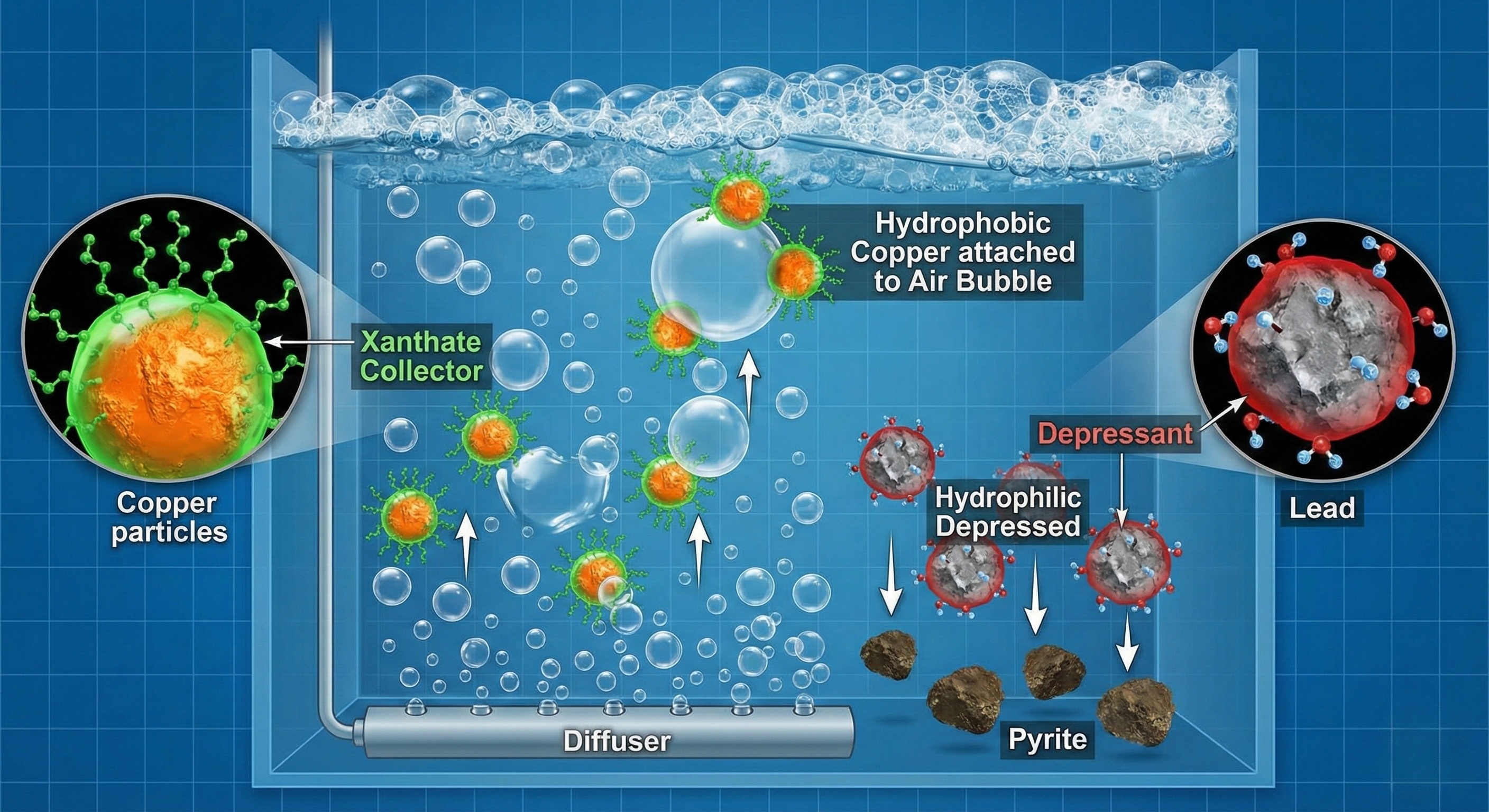
You cannot buy a Flotation Machine and expect it to magically separate metals. The machinery merely provides the mixing and the air bubbles; the reagents do the actual sorting.
Part 5: The Flotation Cell Layout (Rougher, Cleaner, Scavenger)
A copper flotation process is never a single-step event. It requires banks of specialized cells to achieve commercial grades.
- Rougher Cells: The first line. The goal is maximum Recovery. We want to pull out every possible piece of copper/lead, even if it brings some waste rock with it.
- Cleaner Cells: The concentrate from the Roughers is pumped to the Cleaners. The goal here is maximum Grade. We re-float the material, often adding more depressants, to reject the waste rock and achieve the final 25%+ Copper or 50%+ Zinc commercial grade.
- Scavenger Cells: The tailings (waste) from the Roughers are given one last chance. Scavengers use aggressive chemical dosing to catch any valuable minerals that escaped. The concentrate from Scavengers is pumped backwards to the Roughers for another try.
Part 6: Dewatering and Tailings Management
Flotation occurs in a liquid slurry (typically 30% solids). Smelters, however, will not buy wet mud. If your shipped concentrate contains more than 8% to 10% moisture, you pay massive shipping costs for water, and smelters apply "transportable moisture limit" (TML) penalties.
The final concentrates (Cu, Pb, and Zn individually) are first pumped to a High-Efficiency Thickener, which uses gravity and flocculants to increase the solid density to 60%. The thickened slurry is then forced through a Plate and Frame Filter Press under high pressure, squeezing out the water to produce a dry, stackable powder ready for export.
FAQ: Troubleshooting Polymetallic Flotation Plants
A: This means copper is "escaping" the copper circuit and floating in the zinc circuit. This is usually due to insufficient collector dosing in the primary copper roughers, or premature activation. Check your pH levels; copper floats best at pH 8.5-9.5. Also, ensure your grind size is liberating the copper from the zinc.
A: This is the trickiest part of lead zinc separation. The standard method is the "Dichromate Method." You add Sodium Dichromate to the bulk concentrate to strongly depress the Lead (Galena), and then you float the Copper. If environmental regulations ban Dichromate, a Cyanide-Zinc complex method is used to depress Copper and float the Lead.
A: Pyrite floats very easily. To stop it (depress it), you must strictly control your pH using Lime. Pyrite is effectively depressed at a pH of 10.5 to 11.5. If your lime feeder fails and the pH drops to 8, Pyrite will flood your froth launders immediately.
A: Air-Inflated Flotation Machines (like the KYF/XCF series) are currently the industry standard for large modern plants. They use an external blower to force air into the cell, rather than relying on the impeller to suck air in. This provides much finer control over bubble size and froth depth, leading to higher grades and lower energy consumption.
Conclusion: The EPC Advantage in Base Metals
Designing a Copper-Lead-Zinc polymetallic plant is not about buying individual crushers and tanks; it is about engineering a highly sensitive, chemically balanced ecosystem. A minor miscalculation in the grinding circuit or a wrong choice in the depressant strategy will turn a potentially lucrative ore body into a financial disaster.
At OreSolution, we mitigate this risk. From comprehensive bench-scale metallurgical testing and 3D plant design to equipment manufacturing and final commissioning, our turnkey EPC services ensure your Copper Flotation Plant operates at peak commercial efficiency.
Are you developing a complex base metal deposit? Contact OreSolution today to discuss your ore mineralogy and let our engineers design your optimal flowsheet.